 
The line between atoms does not represent a pair of electrons as it does in Lewis diagrams. Valence only describes connectivity, it does not describe the geometry of molecular compounds, or what are now known to be ionic compounds or giant covalent structures. Valence diagrams of a compound represent the connectivity of the elements, lines between two elements, sometimes called bonds, represented a saturated valency for each element. Chlorine, as it has a valence of one, can be substituted for hydrogen, so phosphorus has a valence of 5 in phosphorus pentachloride, PCl 5. 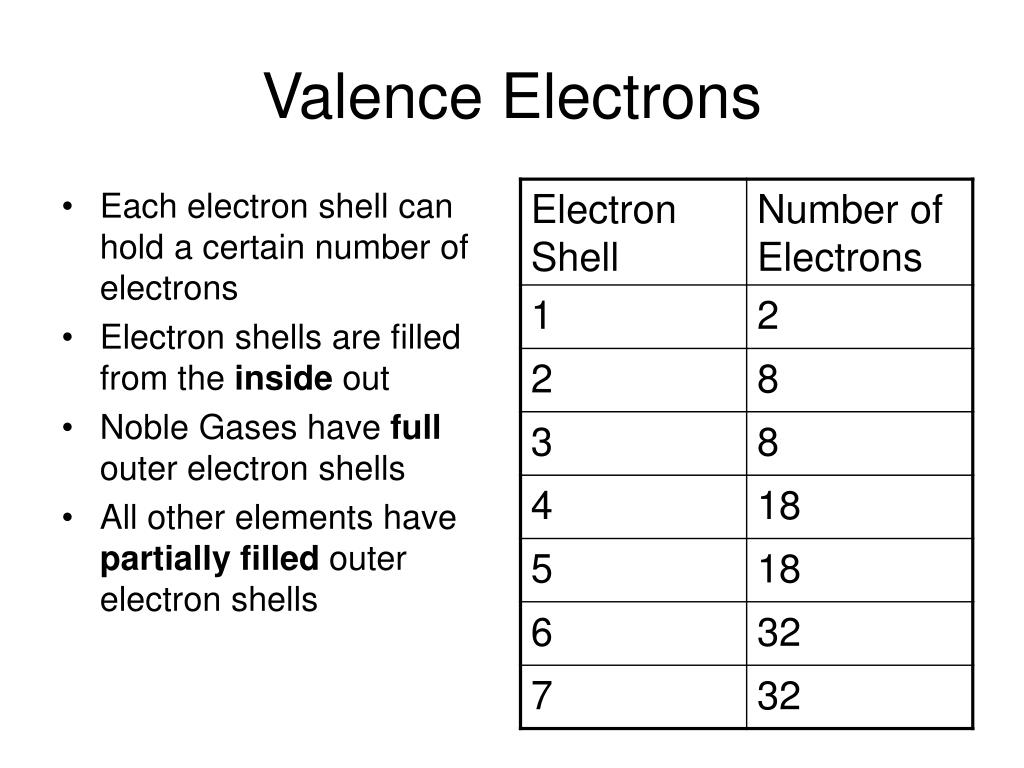
In methane, carbon has a valence of 4 in ammonia, nitrogen has a valence of 3 in water, oxygen has a valence of two and in hydrogen chloride, chlorine has a valence of 1. Silicon has 14 electrons out of which 4 valence electrons are present in the 3s2 3p2 outer orbitals of atom. How many valence electrons does a Silicon atom have Silicon has 4 valence electrons. The combining power or affinity of an atom of an element was determined by the number of hydrogen atoms that it combined with. Who discovered Silicon The element Silicon was discovered by J. The quest for the underlying causes of valence lead to the modern theories of chemical bonding, including Lewis structures (1916), valence bond theory (1927), molecular orbitals (1928), valence shell electron pair repulsion theory (1958) and all the advanced methods of quantum chemistry. The concept of valence was developed in the last half of the 19th century and was successful in explaining the molecular structure of many organic compounds. The valence (or valency) of an element is a measure of its combining power with other atoms when it forms chemical compounds or molecules. In bonding, the valence electrons take part. When an electron loses energy (thereby causing a photon to be emitted), then it can move to an inner shell which is not fully occupied. Valence electrons refer to the electrons present in the outermost or valence shell of an atom of any element. Or the electron can even break free from its associated atom's valence shell this is ionization to form a positive ion. 
An energy gain can trigger an electron to move (jump) to an outer shell this is known as atomic excitation. Like an electron in an inner shell, a valence electron has the ability to absorb or release energy in the form of a photon. An atom with one or two valence electrons fewer than a closed shell is also highly reactive, because of a tendency either to gain the missing valence electrons (thereby forming a negative ion), or to share valence electrons (thereby forming a covalent bond). An atom with one or two valence electrons more than a closed shell is highly reactive, because the extra valence electrons are easily removed to form a positive ion. 
The presence of valence electrons can determine the element's chemical properties and whether it may bond with other elements: For a main group element, a valence electron can only be in the outermost electron shell.Īn atom with a closed shell of valence electrons (corresponding to an electron configuration \(s^2p^6\)) tends to be chemically inert. Also, the first shell gets filled with 2 electrons, the second shell with 8 electrons so remaining four electrons are filled in the outermost third shell.\)Ī valence electron is an electron that is associated with an atom, and that can participate in the formation of a chemical bond in a single covalent bond, both atoms in the bond contribute one valence electron in order to form a shared pair. Oxygen belongs to group 6 and has a valence electron count of 6. Carbon, for instance, belongs to group 4 and has four valence electrons. Draw the Lewis Structure for each element. Number of valence electrons Main group number (neutral atoms) The main group number of an element can be found in its periodic table column. Darken the correct circle to show if the element is a solid (S), liquid (L), or gas (G) at room temperature. Determine the number of protons, neutrons, and electrons in each element. $1$.Valence electrons are electrons present in outermost shell. element’s atomic number, name, and atomic mass. Hint: Silicon belongs to group 14 in periodic table. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
